Atoms
Atoms
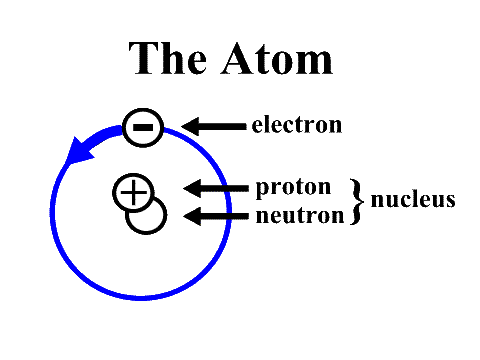
The building blocks of matter are called atoms. Atoms are also a fundamental piece of matter and they are the basic unit of every element in the universe. Matter is anything that can be touched physically. Everything in the universe is made of matter, therefore everything is made of atoms. You may know that everything is made up of molecules, but molecules are made up of atoms, therefore, everything - you, me, everything - is made up of atoms. Water is made up of water molecules. However, water is made up of atoms. There are 2 Hydrogen atoms and 1 Oxygen atom. Therefore, when these atoms join together (H2O), they form a molecule, and when molecules form together, they make something. Atoms are found everyday and can be seen all around us. The word atom is a Greek word, meaning indivisible, basically meaning that it can't be divided. This also means that atoms can't be cut further. For example, if you cut an apple into tiny pieces that they are so small that they cannot be cut any further, they are then indivisible. They are then called atoms, the smallest unit that something can be divided into. Atoms are so small that that the human eye cannot see an atom. They are the smallest unit of everything. Atoms can only be seen using a special microscope. An atom is made up of three tiny particles called subatomic particles. These particles are called protons, neutrons and electrons. Protons are used to calculate the atomic mass of an atom, it also plays a role in determining whether an atom is ion +1 or ion -1. An ion is essentially an electrically charged atom. Ion+ is basically when the positive charge is greater than the negative charge. On the other hand, the exact opposite, ion- is basically when the negative charge is greater than the positive charge. The number of protons in the nucleus determine the atomic number of an element. For example, Hydrogen has an atomic number of 1, therefore it has 1 proton in it's nucleus. Protons and neutrons cluster together in the centre of the atom, forming the nucleus. The electrons fly around and 'orbit' the nucleus in a small cloud. The neutrons, however, hold no charge. The electrons hold a negative charge, while the protons carry a positive charge. That's what keeps the atom together, which is similar to the concept of magnets. The flow of an electron to another electron is electricity. The negative electrons are attracted to the positive nucleus by the same electrical force. Every, and any, element that is listed on the periodic table is consisted of atoms. Some of these elements include hydrogen, helium, oxygen and uranium. These elements are a just a few examples of the many that are listed on the periodic table.
Quarks and Gluons
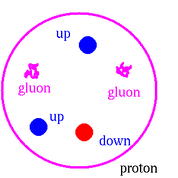
Protons and neutrons are both composed of other particles called quarks and gluons. Protons contain two 'up' quarks and one 'down' quark while neutrons contain one 'up' quark and two 'down' quarks. The gluons are responsible for binding the quarks to one another.
The building blocks of matter are called atoms. Atoms are also a fundamental piece of matter and they are the basic unit of every element in the universe. Matter is anything that can be touched physically. Everything in the universe is made of matter, therefore everything is made of atoms. You may know that everything is made up of molecules, but molecules are made up of atoms, therefore, everything - you, me, everything - is made up of atoms. Water is made up of water molecules. However, water is made up of atoms. There are 2 Hydrogen atoms and 1 Oxygen atom. Therefore, when these atoms join together (H2O), they form a molecule, and when molecules form together, they make something. Atoms are found everyday and can be seen all around us. The word atom is a Greek word, meaning indivisible, basically meaning that it can't be divided. This also means that atoms can't be cut further. For example, if you cut an apple into tiny pieces that they are so small that they cannot be cut any further, they are then indivisible. They are then called atoms, the smallest unit that something can be divided into. Atoms are so small that that the human eye cannot see an atom. They are the smallest unit of everything. Atoms can only be seen using a special microscope. An atom is made up of three tiny particles called subatomic particles. These particles are called protons, neutrons and electrons. Protons are used to calculate the atomic mass of an atom, it also plays a role in determining whether an atom is ion +1 or ion -1. An ion is essentially an electrically charged atom. Ion+ is basically when the positive charge is greater than the negative charge. On the other hand, the exact opposite, ion- is basically when the negative charge is greater than the positive charge. The number of protons in the nucleus determine the atomic number of an element. For example, Hydrogen has an atomic number of 1, therefore it has 1 proton in it's nucleus. Protons and neutrons cluster together in the centre of the atom, forming the nucleus. The electrons fly around and 'orbit' the nucleus in a small cloud. The neutrons, however, hold no charge. The electrons hold a negative charge, while the protons carry a positive charge. That's what keeps the atom together, which is similar to the concept of magnets. The flow of an electron to another electron is electricity. The negative electrons are attracted to the positive nucleus by the same electrical force. Every, and any, element that is listed on the periodic table is consisted of atoms. Some of these elements include hydrogen, helium, oxygen and uranium. These elements are a just a few examples of the many that are listed on the periodic table.
Quarks and Gluons
Protons and neutrons are both composed of other particles called quarks and gluons. Protons contain two 'up' quarks and one 'down' quark while neutrons contain one 'up' quark and two 'down' quarks. The gluons are responsible for binding the quarks to one another.
Isotopes
An isotope is defined as 'each of two or more forms of the same element that contain equal numbers of protons but different numbers of neutrons in their nuclei, and hence differ in relative atomic mass but not in chemical properties; in particular, a radioactive form of an element.'
Ions
An ion is an electrically charged atom. Charged atoms have either too many electrons, or too few electrons. Atoms are heated until they have very high energy, therefore becoming unstable. They are then knocked by electrons that are discharged by a 'cathode ray' in the thruster chamber. When the electrons hit the atoms in the chamber, they cause some of the electrons in the atoms to be forced or stripped away. The mass of the very hot and charged atoms with separated electrons, becomes a 'plasma.'
To make a positive ion, an atom has to become positively charged. This happens when one or more electrons bounce of an atom. On the other hand, negative ions have to become negatively charged. This occurs when an electron is added to an atom.
An isotope is defined as 'each of two or more forms of the same element that contain equal numbers of protons but different numbers of neutrons in their nuclei, and hence differ in relative atomic mass but not in chemical properties; in particular, a radioactive form of an element.'
Ions
An ion is an electrically charged atom. Charged atoms have either too many electrons, or too few electrons. Atoms are heated until they have very high energy, therefore becoming unstable. They are then knocked by electrons that are discharged by a 'cathode ray' in the thruster chamber. When the electrons hit the atoms in the chamber, they cause some of the electrons in the atoms to be forced or stripped away. The mass of the very hot and charged atoms with separated electrons, becomes a 'plasma.'
To make a positive ion, an atom has to become positively charged. This happens when one or more electrons bounce of an atom. On the other hand, negative ions have to become negatively charged. This occurs when an electron is added to an atom.
Non-Atoms
According to http://chemistry.about.com/od/atomicstructure/f/What-Is-An-Atom.htm, some things are not considered atoms. Some matter is either smaller or larger than an atom. Protons, neutrons and electrons are examples of particles that make up atoms but by themselves are not necessarily considered atoms. Molecules and compounds consists of atoms but are not themselves atoms. For example, some molecules and compounds include salt (NaCl), water (H2O) and ethanol (CH2OH). Ions are electrically charged atoms. However, they are still 'types' of atoms. Mono-atomic ions include H+ and O2-. There are also molecular ions, which are not atoms (for example ozone, O3-). Most hydrogen "atoms" do not have a proton, neutron and electron. Given that the number of protons determines the identity of an element, many scientists consider a single proton to be an atom of the element hydrogen.
Types of Atoms
In a normal atom, also known as a neutral atom, there is the same amount of subatomic particles. This means the atom contains the same amount of protons, neutrons and electrons. These neutral atoms include:
According to http://chemistry.about.com/od/atomicstructure/f/What-Is-An-Atom.htm, some things are not considered atoms. Some matter is either smaller or larger than an atom. Protons, neutrons and electrons are examples of particles that make up atoms but by themselves are not necessarily considered atoms. Molecules and compounds consists of atoms but are not themselves atoms. For example, some molecules and compounds include salt (NaCl), water (H2O) and ethanol (CH2OH). Ions are electrically charged atoms. However, they are still 'types' of atoms. Mono-atomic ions include H+ and O2-. There are also molecular ions, which are not atoms (for example ozone, O3-). Most hydrogen "atoms" do not have a proton, neutron and electron. Given that the number of protons determines the identity of an element, many scientists consider a single proton to be an atom of the element hydrogen.
Types of Atoms
In a normal atom, also known as a neutral atom, there is the same amount of subatomic particles. This means the atom contains the same amount of protons, neutrons and electrons. These neutral atoms include:
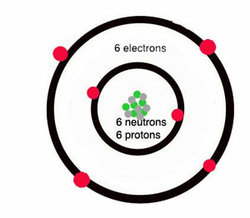
- Carbon Atoms
Carbon is one of the most abundant elements in the universe. In fact, it is the sixth most abundant element in the universe. In addition, carbon is a very special element because it is important in the chemistry of life (to see more information on the chemistry of life, go here). There are 4 known allotropes of carbon, these are amorphous, graphite, diamond and fullerine. Allotropes are the physical forms in which an element can exist. Although the Carbon atom has 6 protons, 6 neutrons and 6 electrons, 4 of the electrons are in the outer shell (valence shell), while its outer shell contains the other 2 electrons. This can be seen in the image above, where the red circles are the electrons, and the green and grey circles are the neutrons and protons.
Basic Information on the Carbon Atom
Source: http://www.chemicalelements.com/elements/c.html
Name: Carbon
Symbol: C
Atomic Number: 6
Atomic Mass: 12.0107 amu
Melting Point: 3500.0 °C (3773.15 K, 6332.0 °F)
Boiling Point: 4827.0 °C (5100.15 K, 8720.6 °F)
Number of Protons/Electrons: 6
Number of Neutrons: 6
Classification: Non-metal
Crystal Structure: Hexagonal
Density @ 293 K: 2.62 g/cm3
Color: Possibly black
Uses of Carbon
Source: http://www.edinformatics.com/math_science/c_atom.htm
Graphite combined with clay form the 'lead' used in pencils.
Diamond is used for decorative purposes, and also as drill bits.
Carbon added to iron makes steel.
Carbon is used for control rods in nuclear reactors.
Graphite carbon in a powdered, caked form is used as charcoal for cooking, artwork and other uses.
Charcoal pills are used in medicine in pill or powder form to adsorb toxins or poisons from the digestive system.
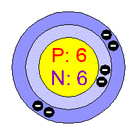
Atomic Structure of Carbon Atoms
Source: http://www.chemicalelements.com/elements/c.html
Number of Energy Levels: 2
First Energy Level: 2
Second Energy Level: 4
Source: http://www.chemicalelements.com/elements/c.html
Number of Energy Levels: 2
First Energy Level: 2
Second Energy Level: 4
Quick Facts about Carbon Atoms
Source: http://www.chemicalelements.com/elements/mg.html
Date of Discovery: Known to the ancients
Discoverer: Unknown
Name Origin: From the Latin carbo (coal)
Uses: steel, filters
Obtained From: burning with insufficient oxygen
Isotopes of Carbon Atoms
Source: http://www.chemicalelements.com/elements/c.html
Isotope Half Life
C-11 20.3 minutes
C-12 Stable
C-13 StableC-145730.0 years
C-15 2.5 seconds
Source: http://www.chemicalelements.com/elements/mg.html
Date of Discovery: Known to the ancients
Discoverer: Unknown
Name Origin: From the Latin carbo (coal)
Uses: steel, filters
Obtained From: burning with insufficient oxygen
Isotopes of Carbon Atoms
Source: http://www.chemicalelements.com/elements/c.html
Isotope Half Life
C-11 20.3 minutes
C-12 Stable
C-13 StableC-145730.0 years
C-15 2.5 seconds
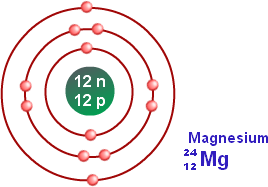
- Magnesium Atoms
There are no allotropes of magnesium. Magnesium is an alkaline earth metal, making up 13% of the planet's mass and a large fraction of the planet's mantle. Magnesium forms a variety of industrially and biologically important compounds, including magnesium oxide, various salts, and others. The history of magnesium is quite interesting. In 1618, a farmer at Epsom in England decided to give his cows water from a well. The cows did not like the water because it had a bitter taste to it, so the cows refused to drink it. The farmer noticed that the water actually healed scratches, rashes, etc. From that day, the substance was known as the Epsom salt, and became very widespread and famous throughout the world. Today, we recognise this to be hydrated magnesium sulfate (MgSO4.7H20).
Basic Information on Magnesium Atoms
Source: http://www.chemicalelements.com/elements/mg.html
Name: Magnesium
Symbol: Mg
Atomic Number: 12
Atomic Mass: 24.305 amu
Melting Point: 650.0 °C (923.15 K, 1202.0 °F)
Boiling Point: 1107.0 °C (1380.15 K, 2024.6 °F)
Number of Protons/Electrons: 12
Number of Neutrons: 12
Classification: Alkaline Earth
Crystal Structure: Hexagonal
Density @ 293 K: 1.738 g/cm3
Color: grayish
Uses of Magnesium
Source: http://www.usesof.net/uses-of-magnesium.html
When burned, magnesium produces a very bright white light, making it ideal for firework sparklers, flares and flash photography.
Functions as an agent to produce uranium out of salt.
The element’s ions are necessary for every living organism.
Magnesium salt is included in fertilizers and food.
Magnesium bromide is utilized as a sedative.
Bromine produces the sedative effect.
Athletes use magnesium carbonate to enhance their grip.
Sulfate is required to make paper.
Three of its compounds are found in antiseptics.
Other compounds are used to make textiles invulnerable to moth.
Due to its light weight and strength it is used in many vehicle parts (to make trucks, automobiles, etc).
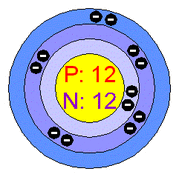
Atomic Structure of Magnesium Atoms
Source: http://www.chemicalelements.com/elements/mg.html
Number of Energy Levels: 3
First Energy Level: 2
Second Energy Level: 8
Third Energy Level: 2
Source: http://www.chemicalelements.com/elements/mg.html
Number of Energy Levels: 3
First Energy Level: 2
Second Energy Level: 8
Third Energy Level: 2
Quick Facts on Magnesium Atoms
Source: http://www.chemicalelements.com/elements/mg.html
Date of Discovery: 1808
Discoverer: Sir Humphrey Davy
Name Origin: Magnesia (City)
Uses: airplanes, missiles
Obtained From: sea water
Isotopes of Magnesium Atoms
Source: http://www.chemicalelements.com/elements/mg.html
Isotope Half Life
Mg-24 Stable
Mg-25 Stable
Mg-26 Stable
Mg-27 9.45 minutes
Mg-28 21.0 hours
Source: http://www.chemicalelements.com/elements/mg.html
Date of Discovery: 1808
Discoverer: Sir Humphrey Davy
Name Origin: Magnesia (City)
Uses: airplanes, missiles
Obtained From: sea water
Isotopes of Magnesium Atoms
Source: http://www.chemicalelements.com/elements/mg.html
Isotope Half Life
Mg-24 Stable
Mg-25 Stable
Mg-26 Stable
Mg-27 9.45 minutes
Mg-28 21.0 hours
Watch this video to learn more about atoms.
Watch this second video to learn even more about atoms.