Elements
Elements found in Fireworks
There are several elements that are used in fireworks. These elements help fireworks do special things. They can help produce a specific colour, they can help create sparkle and glitter effects, they can act as oxidizers, they can do anything that can help any fireworks show flow smoothly. The following are all the elements that are listed on the Periodic Table that are involved in creating the perfect fireworks show:
There are several elements that are used in fireworks. These elements help fireworks do special things. They can help produce a specific colour, they can help create sparkle and glitter effects, they can act as oxidizers, they can do anything that can help any fireworks show flow smoothly. The following are all the elements that are listed on the Periodic Table that are involved in creating the perfect fireworks show:
- Aluminum - Aluminum is used to produce silver and white flames and sparks. It is a common component of sparklers.
- Antimony - Antimony is used to create firework glitter effects.
- Barium - Barium is used to create green colors in fireworks and it can also help stabilize other volatile elements.
- Calcium - Calcium is used to deepen firework colors. Calcium salts produce orange fireworks.
- Carbon - Carbon is one of the main components of black powder, which is used as a propellent in fireworks. Carbon provides the fuel for a firework. Common forms include carbon black, sugar, or starch.
- Chlorine - Chlorine is an important component of many oxidizers in fireworks. Several of the metal salts that produce colors contain chlorine.
- Copper - Copper compounds produce blue colors in fireworks.
- Iron - Iron is used to produce sparks. The heat of the metal determines the color of the sparks.
- Lithium - Lithium is a metal that is used to impart a red color to fireworks. Lithium carbonate, in particular, is a common colorant.
- Magnesium - Magnesium burns a very bright white, so it is used to add white sparks or improve the overall brilliance of a firework.
- Oxygen - Fireworks include oxidizers, which are substances that produce oxygen in order for burning to occur. The oxidizers are usually nitrates, chlorates, or perchlorates. Sometimes the same substance is used to provide oxygen and color.
- Phosphorus - Phosphorus burns spontaneously in air and is also responsible for some glow-in-the-dark effects. It may be a component of a firework's fuel.
- Potassium - Potassium helps to oxidize firework mixtures. Potassium nitrate, potassium chlorate, and potassium perchlorate are all important oxidizers.
- Sodium - Sodium imparts a gold or yellow color to fireworks, however, the color may be so bright that it masks less intense colors.
- Sulfur - Sulfur is a component of black powder. It is found in a firework's propellant/fuel.
- Strontium - Strontium salts impart a red color to fireworks. Strontium compounds are also important for stabilizing fireworks mixtures.
- Titanium - Titanium metal can be burned as powder or flakes to produce silver sparks.
- Zinc - Zinc is used to create smoke effects for fireworks and other pyrotechnic devices.
What are Elements?
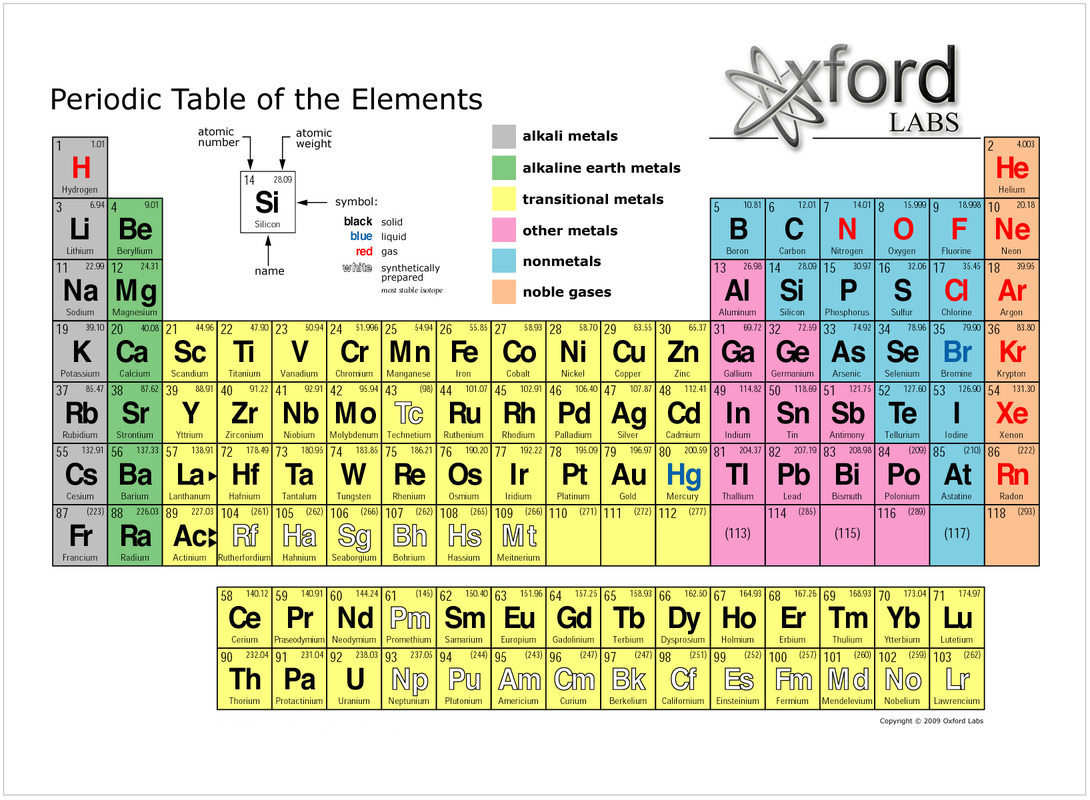
Elements are made by the number of subatomic particles - protons, neutrons and electrons - that are held together. Elements are divided into 8 categories. These are alkali metals, alkaline earth metals, transition metals, other metals, metalloids, other non metals, halogens and noble gas. All elements have melting points and boiling points. Some elements have a very low boiling point. For example, hydrogen has a boiling point of -252.9 degrees Celsius. On the other hand, some elements have a very high boiling point. For example, hafnium has a boiling point of 4,602.9 degrees Celsius. Currently, there are 118 elements that have been discovered. These elements are displayed on the periodic table. The periodic table gives the name, symbol (for example, H for Hydrogen), the atomic number (for example, Hydrogen has an atomic number of 1, as shown in the periodic table above), the chemical properties and the electron configurations of each element. An element is made up of molecules. Molecules are slightly larger than atoms. They are created when atoms form a bond together, however, they are still too small to see with our eyes. A popular example of an element is Hydrogen. Hydrogen is the first element on the periodic table. It has an atomic mass of 1 (protons + neutrons). Hydrogen physically looks like gas or water, depending on the temperature. If the temperature is hot enough, the gas turns into water. Hydrogen has a symbol of H. These symbols can be used to represent chemical formulas. For example, the chemical formula for water is H2O. This basically means 2 Hydrogen atoms + 1 Oxygen atom makes water. The atomic mass of hydrogen is close to, or exactly 1.00794, therefore, it is the lightest element in the periodic table. Hydrogen is very abundant. It makes up to 75% of the element mass of the universe. Hydrogen has many uses. It also can be combined with other elements for various purposes.
Molecules
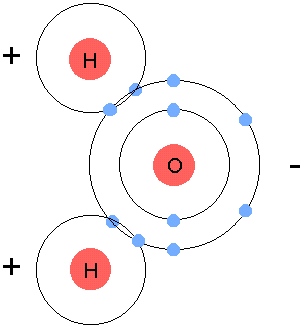
Molecules are basically when two or more atoms join together. Molecules are basically atoms on a larger scale. If you break up molecules, you get atoms, therefore everything is made up of molecules and atoms. A molecule can be anything that you think of - a tree, to a pen. When atoms join to form molecules, they are held together with a chemical bond. These chemical bonds form because of sharing or exchanging electrons between atoms. However, the only electrons that are shared are in the outer shell (valence shell). The bond that the atoms share are pretty simple. For example, a water molecule is made up of 3 atoms - 2 Hydrogen atoms, and 1 Oxygen atom. The Hydrogen atom has 1 electron in it's valence shell, however it always looks for another electron to fill its maximum capacity of electrons, which is two. On the other hand, Oxygen atoms can have up 8 electrons in it's valence shell. When the oxygen atom has 6 electrons, it looks for 2 more electrons to share. This is when the bond comes in. The 2 Hydrogen atoms connect to the oxygen atom, sharing one atom to each other. One hydrogen atom connects to the oxygen atom. When this occurs, the hydrogen atom shares one electron to the oxygen atom, and the oxygen atom shares one electron to each of the hydrogen atoms, filling each others maximum capacity of electrons. The other hydrogen atom does the exact same as the other one. As seen in the image below, the hydrogen atoms each have 2 electrons as the oxygen atom shares 1 electron to each of the hydrogen atoms. The oxygen atom has 8 electrons, because the hydrogen atoms each share 1 atom, and since there is 2 hydrogen atoms in a water molecule, both of them share a total of 2 atoms, making the oxygen atom have from 6 electrons, to 8 electrons.
Elements are made by the number of subatomic particles - protons, neutrons and electrons - that are held together. Elements are divided into 8 categories. These are alkali metals, alkaline earth metals, transition metals, other metals, metalloids, other non metals, halogens and noble gas. All elements have melting points and boiling points. Some elements have a very low boiling point. For example, hydrogen has a boiling point of -252.9 degrees Celsius. On the other hand, some elements have a very high boiling point. For example, hafnium has a boiling point of 4,602.9 degrees Celsius. Currently, there are 118 elements that have been discovered. These elements are displayed on the periodic table. The periodic table gives the name, symbol (for example, H for Hydrogen), the atomic number (for example, Hydrogen has an atomic number of 1, as shown in the periodic table above), the chemical properties and the electron configurations of each element. An element is made up of molecules. Molecules are slightly larger than atoms. They are created when atoms form a bond together, however, they are still too small to see with our eyes. A popular example of an element is Hydrogen. Hydrogen is the first element on the periodic table. It has an atomic mass of 1 (protons + neutrons). Hydrogen physically looks like gas or water, depending on the temperature. If the temperature is hot enough, the gas turns into water. Hydrogen has a symbol of H. These symbols can be used to represent chemical formulas. For example, the chemical formula for water is H2O. This basically means 2 Hydrogen atoms + 1 Oxygen atom makes water. The atomic mass of hydrogen is close to, or exactly 1.00794, therefore, it is the lightest element in the periodic table. Hydrogen is very abundant. It makes up to 75% of the element mass of the universe. Hydrogen has many uses. It also can be combined with other elements for various purposes.
Molecules
Molecules are basically when two or more atoms join together. Molecules are basically atoms on a larger scale. If you break up molecules, you get atoms, therefore everything is made up of molecules and atoms. A molecule can be anything that you think of - a tree, to a pen. When atoms join to form molecules, they are held together with a chemical bond. These chemical bonds form because of sharing or exchanging electrons between atoms. However, the only electrons that are shared are in the outer shell (valence shell). The bond that the atoms share are pretty simple. For example, a water molecule is made up of 3 atoms - 2 Hydrogen atoms, and 1 Oxygen atom. The Hydrogen atom has 1 electron in it's valence shell, however it always looks for another electron to fill its maximum capacity of electrons, which is two. On the other hand, Oxygen atoms can have up 8 electrons in it's valence shell. When the oxygen atom has 6 electrons, it looks for 2 more electrons to share. This is when the bond comes in. The 2 Hydrogen atoms connect to the oxygen atom, sharing one atom to each other. One hydrogen atom connects to the oxygen atom. When this occurs, the hydrogen atom shares one electron to the oxygen atom, and the oxygen atom shares one electron to each of the hydrogen atoms, filling each others maximum capacity of electrons. The other hydrogen atom does the exact same as the other one. As seen in the image below, the hydrogen atoms each have 2 electrons as the oxygen atom shares 1 electron to each of the hydrogen atoms. The oxygen atom has 8 electrons, because the hydrogen atoms each share 1 atom, and since there is 2 hydrogen atoms in a water molecule, both of them share a total of 2 atoms, making the oxygen atom have from 6 electrons, to 8 electrons.
Types of Chemical Bonds
Depending on the element, the electrons form one of the different type of chemical bonds. There are various chemical bonds that are different, according to the elements that are connected. These types of bonds are known as:
Depending on the element, the electrons form one of the different type of chemical bonds. There are various chemical bonds that are different, according to the elements that are connected. These types of bonds are known as:
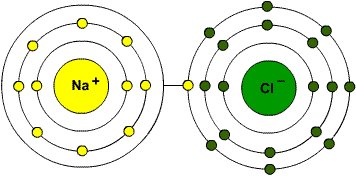
- Ionic bonds
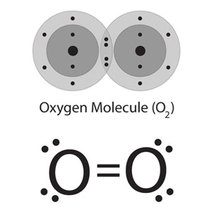
- Covalent bonds
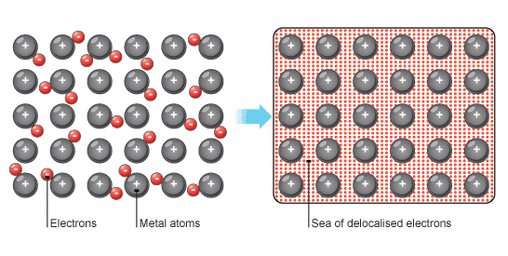
- Metallic bonds
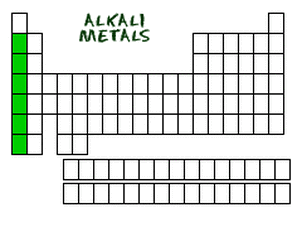
Alkali Metals
Source: http://www.chemicalelements.com/groups/alkali.html
The alkali metals, found in group 1 of the periodic table (also known as group IA), are known to be very reactive. They do not occur freely in nature. These metals only have one electron in it's valence shell (outer shell). This means that these elements are ready to lose that electron when going through an ionic bond. Just like all metals, alkali metals are workable, mouldable, flexible and are great conductors of heat and electricity. However, alkali metals are still different to most other metals. They are softer and react very easily. The most reactive elements in this group are cesium and francium, as just like all alkali metals, can explode if exposed to water.
The Alkali Metals are:
Watch this video to see what happens when potassium reacts to water.
Source: http://www.chemicalelements.com/groups/alkali.html
The alkali metals, found in group 1 of the periodic table (also known as group IA), are known to be very reactive. They do not occur freely in nature. These metals only have one electron in it's valence shell (outer shell). This means that these elements are ready to lose that electron when going through an ionic bond. Just like all metals, alkali metals are workable, mouldable, flexible and are great conductors of heat and electricity. However, alkali metals are still different to most other metals. They are softer and react very easily. The most reactive elements in this group are cesium and francium, as just like all alkali metals, can explode if exposed to water.
The Alkali Metals are:
- Lithium
- Sodium
- Potassium
- Rubidium
- Cesium
- Francium
Watch this video to see what happens when potassium reacts to water.
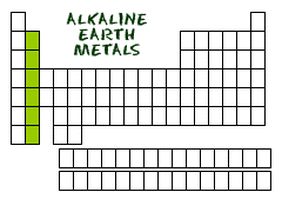
Alkaline Earth Metals
Source: http://www.chemicalelements.com/groups/alkaline.html
The alkaline earth elements are metallic elements found in the second group of the periodic table. All alkaline earth elements have an oxidation number of +2, making them very reactive. Because of their reactivity, the alkaline metals are not found free in nature. All of the Alkaline Earth Metals are all elements with a shiny, silvery-white colour. These elements are all found in the Earth's crust, but in a different form as they are very reactive. They are usually distributed in rock structures. Magnesium is an Alkaline Earth Metal, and is found in minerals including carnellite, dolomite and magnesite. Calcium is also an Alkaline Earth Metal and is found in chalk, limestone, gypsum and anhydrite. Magnesium is the eighth most abundant element in the Earth's crust, while calcium is the fifth. The metals of Group 2 are harder and denser than sodium and potassium, and have higher melting points. These properties are due largely to the presence of two valence electrons on each atom, which leads to stronger metallic bonding than occurs in Group 1.
The Alkaline Earth Metals are:
Watch this video to see the group 2 elements.
Source: http://www.chemicalelements.com/groups/alkaline.html
The alkaline earth elements are metallic elements found in the second group of the periodic table. All alkaline earth elements have an oxidation number of +2, making them very reactive. Because of their reactivity, the alkaline metals are not found free in nature. All of the Alkaline Earth Metals are all elements with a shiny, silvery-white colour. These elements are all found in the Earth's crust, but in a different form as they are very reactive. They are usually distributed in rock structures. Magnesium is an Alkaline Earth Metal, and is found in minerals including carnellite, dolomite and magnesite. Calcium is also an Alkaline Earth Metal and is found in chalk, limestone, gypsum and anhydrite. Magnesium is the eighth most abundant element in the Earth's crust, while calcium is the fifth. The metals of Group 2 are harder and denser than sodium and potassium, and have higher melting points. These properties are due largely to the presence of two valence electrons on each atom, which leads to stronger metallic bonding than occurs in Group 1.
The Alkaline Earth Metals are:
- Beryllium
- Magnesium
- Calcium
- Strontium
- Barium
- Radium
Watch this video to see the group 2 elements.
Watch this video to learn more about the Periodic Table of Elements.