Reactions
Reactions
A chemical reaction is when a chemical substance transforms into another. There are 4 types of chemical reactions. These types of reactions are common, they are a combination reaction, a decomposition reaction, a precipitation reaction and a combustion reaction.
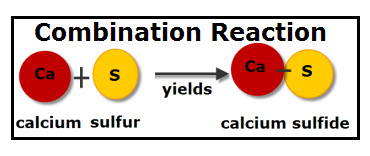
A combination reaction requires one or more substance reacting together to create a new substance. A popular example of a combination reaction is water. Water is created when two hydrogen atoms and one oxygen atom bond together (H2+O ---> H2O). The substances that are combining to make this a combination reaction are hydrogen and oxygen. These two elements join together to create a new substance, in this case, water. Water is a different substance than hydrogen and oxygen. On the other hand, ozone (O3) is a reaction, however it doesn't combine to create a new substance. When they combine, it is still made up of the same thing.
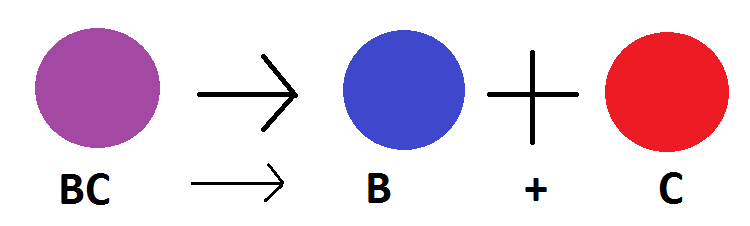
The decomposition reaction is when a single substance reacts and breaks down or 'decomposes' into new substances. For example, carbonic acid canbe broken down into water and carbon dioxide. The chemical formula for this reaction is H2CO3 (carbonic acid) --> H2O (water) + CO2 (carbon dioxide). In addition, water can be further broken down into more substances, causing a 'chain-decomposition reaction,' so to speak. Water can be further decomposed into hydrogen and oxygen gas. This occurs when and electrical current passes through the water. The chemical formula for this decomposition reaction is H2O (water) --> H2 (hydrogen) + O2 (oxygen).
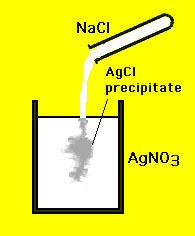
A precipitation reaction requires a substance to be dissolved in water. A soluble is a substance that be dissolved in water. On the other hand, an insoluble substance are substances that cannot be dissolved in water. Therefore, a precipitation reaction can only occur with soluble substances. In a precipitation reaction, two soluble substances combine in water and forms a new insoluble substance. Therefore, it can be said that when an insoluble substance is formed, it is called precipitation. The insoluble substance should separate from water, making it appear cloudy. After a while, the substance sinks to the bottom of a container, therefore leaving a clear solution. The insoluble substance that is formed after the precipitation reaction is known as the precipitate. For example, calcium hydroxide solution and carbon dioxide makes an insoluble substance called calcium carbonate + water. This is a precipitation reaction because calcium hydroxide and carbon dioxide are both soluble substances. Once they are combined, they create an insoluble precipitate.
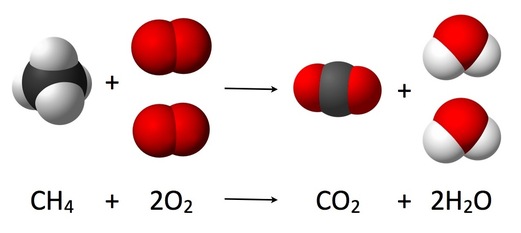
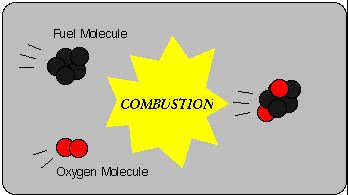
A combustion reaction happens when a substance reacts to oxygen and releases energy. Fireworks use this reaction when potassium nitride realeases oxygen after being heated and combined with charcoal and carbon. This then creates nitrogen gas. The explosion is then created when nitrogen expands and carbon puts pressure on the container. This is very important otherwise the fireworks will not work. Once energy is released, a new substances is formed. With this new substance, heat or light is present. Basically, a combustion reaction occurs when something burns or explodes. An experiment that can be done to see a combustion reaction is when a magnesium ribbon is burned. Oxygen combines with the magnesium oxide (magnesium metal + oxygen gas --> magnesium oxide). Magnesium oxide is created when oxygen combines with the metal to produce a white powder. Combustion reactions are very important as it is used to power car engines, to produce energy and through many more uses.
A chemical reaction is when a chemical substance transforms into another. There are 4 types of chemical reactions. These types of reactions are common, they are a combination reaction, a decomposition reaction, a precipitation reaction and a combustion reaction.
A combination reaction requires one or more substance reacting together to create a new substance. A popular example of a combination reaction is water. Water is created when two hydrogen atoms and one oxygen atom bond together (H2+O ---> H2O). The substances that are combining to make this a combination reaction are hydrogen and oxygen. These two elements join together to create a new substance, in this case, water. Water is a different substance than hydrogen and oxygen. On the other hand, ozone (O3) is a reaction, however it doesn't combine to create a new substance. When they combine, it is still made up of the same thing.
The decomposition reaction is when a single substance reacts and breaks down or 'decomposes' into new substances. For example, carbonic acid canbe broken down into water and carbon dioxide. The chemical formula for this reaction is H2CO3 (carbonic acid) --> H2O (water) + CO2 (carbon dioxide). In addition, water can be further broken down into more substances, causing a 'chain-decomposition reaction,' so to speak. Water can be further decomposed into hydrogen and oxygen gas. This occurs when and electrical current passes through the water. The chemical formula for this decomposition reaction is H2O (water) --> H2 (hydrogen) + O2 (oxygen).
A precipitation reaction requires a substance to be dissolved in water. A soluble is a substance that be dissolved in water. On the other hand, an insoluble substance are substances that cannot be dissolved in water. Therefore, a precipitation reaction can only occur with soluble substances. In a precipitation reaction, two soluble substances combine in water and forms a new insoluble substance. Therefore, it can be said that when an insoluble substance is formed, it is called precipitation. The insoluble substance should separate from water, making it appear cloudy. After a while, the substance sinks to the bottom of a container, therefore leaving a clear solution. The insoluble substance that is formed after the precipitation reaction is known as the precipitate. For example, calcium hydroxide solution and carbon dioxide makes an insoluble substance called calcium carbonate + water. This is a precipitation reaction because calcium hydroxide and carbon dioxide are both soluble substances. Once they are combined, they create an insoluble precipitate.
A combustion reaction happens when a substance reacts to oxygen and releases energy. Fireworks use this reaction when potassium nitride realeases oxygen after being heated and combined with charcoal and carbon. This then creates nitrogen gas. The explosion is then created when nitrogen expands and carbon puts pressure on the container. This is very important otherwise the fireworks will not work. Once energy is released, a new substances is formed. With this new substance, heat or light is present. Basically, a combustion reaction occurs when something burns or explodes. An experiment that can be done to see a combustion reaction is when a magnesium ribbon is burned. Oxygen combines with the magnesium oxide (magnesium metal + oxygen gas --> magnesium oxide). Magnesium oxide is created when oxygen combines with the metal to produce a white powder. Combustion reactions are very important as it is used to power car engines, to produce energy and through many more uses.
Here are some images on each of these 4 types of chemical reactions.
An example of a combination reaction is:
An example of a combination reaction is:
An example of a decomposition reaction is:
An example of a precipitation reaction is:
An example of a combustion reaction is:
Watch this video to learn more about the types of chemical reactions.